Understanding the differences between pool and hot tub water quality
Chlorine
Chlorine is clearly the preferred sanitizer/oxidizer product in the pool/hot tub industry, as it is readily available in a variety of forms (e.g. powder, granules, liquid, tablets, pucks, etc.). The primary question to ask before using chlorine is whether the hot tub is indoors or outdoors. If indoors, then chlorine is not always the best choice since it can create a multitude of problems if the hot tub is not properly maintained. The poor air circulation combined with a buildup of combined chlorine can cause irritation to the eyes and mucous membranes and also create that smelly ‘chlorine odour’ which is often associated with indoor hot tubs.
If the hot tub is located outside and the customer prefers to use chlorine, remember to use a ‘stabilized’ form (e.g. dichlor or trichlor), which will slow down the decomposition of the chorine due to sunlight.
Dichlor is almost pH neutral at 6.6, while Trichlor carries an acidic pH (2.8 to 3.5). The unstabilized forms of chlorine (e.g. calcium, lithium, and sodium) all have higher pH levels ranging from 8.5 to 13. Understanding the pH levels of products will help to better control the water’s pH levels.
Bromine
There are four key advantages of using bromine over chlorine in hot tub environments:
- It is more effective over a wider pH range, whereas chlorine’s effectiveness decreases as pH rises.
- Bromamines are almost as effective at sanitizing/oxidizing as free bromine.
- Bromamines, formed in reaction with ammonia wastes, do not have the unpleasant odour of chloramines and are less irritating to the eyes and respiratory tract.
- Bromine lasts longer in hot water environments than unstabilized chlorine.
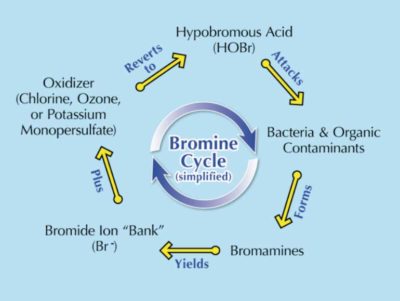
Generally, bromine can be purchased in tablet form: BCDMH (1-Bromo-3-chloro-5,5-dimethylhydantoin) or DBDMH (1,3-Dibromo-5,5-dimethylhydantoin). Tablets are placed in an erosion feeder (or floater), and as hot tub water spills over the tablets, bromine is introduced into the water. Bromine is unique in that it needs a ‘kick start’ to get it going (like starting a motorcycle or dirt bike). This can be in the form of any oxidizing substance (e.g. chlorine, ozone, or monopersulfate). This in turn starts what is called the ‘bromine cycle’ (see Figure 1).
If BCDMH tablets are used there is no need to add a separate oxidizer—it is already there (the ‘C’ in BCDMH stands for chlorine). If using DBDMH, an oxidizer will need to be added separately.
The one issue with using bromine is that it is slightly acidic. BCDMH has a pH of 4.5 to 4.8 and DMDBH has a pH of 6.6. These lower pH levels can be compensated by following proper water balance practices—specifically, making sure the TA level is sufficiently high (but still within acceptable ranges) to offset the lower pH. Hot tub equipment can be damaged if exposed to low pH levels.
Another important factor to consider is bromine has no commercially available stabilizer; therefore, it is best used for indoor hot tubs or units installed outdoors with a cover.
Ozone (O3)
Ozone is another option for sanitizing hot tubs. Gaining in popularity, many hot tub manufacturers now include ozonators in their units. Ozone is an excellent oxidizer, but its usefulness as a sanitizer is limited as it readily converts to oxygen as soon as it hits the water’s surface. If using ozone, manufacturers recommend using a minute amount of chlorine or bromine to create a sanitizing residual in the water—usually half of what would be used if no ozone were present.







